
Concept explainers
a)
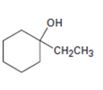
Interpretation:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how 1-ethylcyclohexanol can be prepared is to be stated.
Concept introduction:
An alicyclic 30 alcohol is to be prepared from benzene. For this purpose benzene has to be converted into an alicyclic
To state:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how to prepare 1-ethylcyclohexanol.
b)
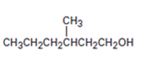
Interpretation:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how 3-methylhexan-1-ol can be prepared is to be given.
Concept introduction:
A six carbon straight chain alcohol with a methyl group on C3 is to be prepared from benzene. By ozonolysis, the benzene ring is broken to get the open chain dialdehyde glyoxal. To one of the aldehydic group in glyoxal, an isopentyl group can be introduced by treating with a suitable Grignard reagent. Upon heating the alcohol will eliminate water to yield an unsaturated
To give:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how to prepare 3-methylhexan-1-ol.
c)
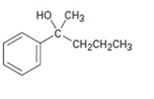
Interpretation:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how 2-methyl-2-phenyl-2 butanol can be prepared is to be given.
Concept introduction:
An
To give:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how to prepare .
d)
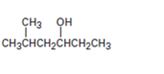
Interpretation:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how 5-methylhexan-3-ol can be prepared is to be given.
Concept introduction:
A six carbon straight chain aldehyde with a methyl group on C4 is to be prepared from benzene. By ozonolysis, the benzene ring is broken to get the open chain dialdehyde, maloaldehyde with three carbon atoms. To one of the aldehydic group in the dialdehyde, an isobutyl group can be introduced by treating with a suitable Grignard reagent to get an aldol. The aldehyde group in the aldol is reduced to an alkyl group to get the alcohol requireds.
To give:
Starting with benzene and using other alcohols of six or fewer carbons as the organic reagents how to prepare 3-methylhexan-1-ol.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardAn important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and its alkyl-substituted derivative: Base CH;CH2O OCH,CH3 CH;CH,0 OCH2CH3 H2 Diethyl malonate Base CH;CH,0 °C `OCH,CH3 CH;CH,O OCH,CH3 R Alkyl substituted diethyl malonate NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is typically used to deprotonate the alkyl-substituted derivative. Explain why.arrow_forwardCCH H20, H2SO4 H9SO4 CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Hjö: -CH3 -CH3 H3O*arrow_forward
- Synthesize each compound from benzonitrile (C6H5CN) as the only organic starting material; that is, every carbon in the product must originate in benzonitrile.arrow_forwardExplain how you would synthesize the following compounds. As raw materials you can use any alcohols containing four or fewer carbon atoms, cyclohexanol, and any necessary inorganic solvents and reagents.arrow_forwardShow how lidocaine, one of the most widely used injectable anesthetics, can be prepared from benzene and compounds containing no more than fourcarbons.arrow_forward
- A postgraduate student wanted to synthesized two carbonyl compounds known as compounds M and N using oxidation of alcohol. However, these carbonyl compounds should have more than 6 carbon atoms Draw two (2) possible structural formulae for compounds M and N. How would you distinguish between M and N?arrow_forwardA postgraduate student wanted to synthesized two carbonyl compounds known as compounds M and N using oxidation of alcohol. However, these carbonyl compounds should have more than 6 carbon atoms. Draw two (2) possible structural formulae for compounds M and N. How would you distinguish between compounds M and N?arrow_forwardFollowing is the structural formula of the tranquilizer meparfynol (Oblivon). Oblivon HO Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains al- kyne and hydroxyl functional groups.)arrow_forward
- Predict the products or starting material of the following reactions. Only the organic products are needed (no byproducts).arrow_forwardDraw the structure of the starting material needed to make 2-methylhept-3-yne using sodium amide in liquid ammonia, followed by 1-bromopropane. The starting hydrocarbon must have no more than five carbons 1) NaNH2, NH3() NaBr + 2) CHзCH2CH2Brarrow_forward13. Synthesize the following compounds using materials with no more than six carbons. to H₂C OH SO3Harrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
