
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7.1, Problem 7.1P
Problem 7-1
In the reaction
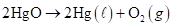
we measure the evolution of oxygen gas to determine the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
HARD
Consider the following reaction and experimental data:
X + Y → XY
Trial
1
2
3
[X] (mol/L) [Y] (mol/L) Rate (molL-¹'s-¹)
0.6
1.8
0.6
0.3
0.3
0.6
1.8
5.4
7.2
FIRST TRY
Using the information above, determine the rate of this reaction when [X] =
1.0 mol/L and [Y] = 2.0 mol/L.
Rate = moll-¹5-1
IL Evaluation
Activity 1: Identification
Directions: Predict the rate of the reaction in the following. Assume that the substances below are
reactants for different reactions. Indicate if the reaction will be slower, faster, or have the
same rate. The first one is done for you.
Normal condition
Changed condition
Factor
Reaction rate
20% HCI
5% HCI
concentration
faster
1. 276 K
315 K
2. A cube of sugar
3. 5 sampalok cube
4. 45 g of Na0H
A powder of sugar
1 sampalok cube
52 g of NaOH
5. 3.68 x 1025 atoms of Li
9.25 x 1023 atoms Li
6. 500 K, 4.5% Barium
600K, 4.5% Barium
7. 45 g.of NaOH
8.560 mL of soy sauce
52 g of NaOH
5.6 L of soy sauce
9.500 K, 4.5% Barium
600K, 5.5% Barium
Starch + water + amylase
10.Starch + water
I Review | Constants | Periodic Table
The half-life of a reaction, t1/2, is the time it takes
for the reactant concentration A to decrease by
half. For example, after one half-life the
concentration falls from the initial concentration
Ao to [Alo/2, after a second half-life to [A]o/4,
after a third half-life to [Alo/8, and so on. on.
v Correct
Part B
A certain second-order reaction (B→products) has a rate constant of 1.80x10-3 M.s-1 at 27°C and an
initial half-life of 246 s. What is the concentration of the reactant B after one half-life?
Express your answer with the appropriate units.
• View Available Hint(s)
?
HA
2.04
M
Chapter 7 Solutions
Introduction to General, Organic and Biochemistry
Ch. 7.1 - Problem 7-1 In the reaction we measure the...Ch. 7.4 - Problem 7-2 Calculate the rate for the reaction in...Ch. 7.6 - Prob. 7.3PCh. 7.6 - Prob. 7.4PCh. 7.6 - Prob. 7.5PCh. 7.7 - Prob. 7.6PCh. 7.7 - Problem 7-7 Consider the following equilibrium...Ch. 7.7 - Prob. 7.8PCh. 7.7 - Prob. 7.9PCh. 7 - 7-10 The rate of disappearance of HCI was measured...
Ch. 7 - 7-11 Consider the following reaction: Suppose we...Ch. 7 - 7-12 Two kinds of gas molecules are reacted at a...Ch. 7 - 7-13 Why are reactions between ions in aqueous...Ch. 7 - Prob. 7.14PCh. 7 - 7-15 A certain reaction is exothermic by 9...Ch. 7 - 7-16 A quart of milk quickly spoils if left at...Ch. 7 - 7-17 If a certain reaction takes 16 h to go to...Ch. 7 - Prob. 7.18PCh. 7 - Prob. 7.19PCh. 7 - Prob. 7.20PCh. 7 - Prob. 7.21PCh. 7 - 7-22 If you add a piece of marble, CaCO3 to a 6 M...Ch. 7 - Prob. 7.23PCh. 7 - Prob. 7.24PCh. 7 - Prob. 7.25PCh. 7 - 7-26 Write the chemical equations corresponding to...Ch. 7 - Prob. 7.27PCh. 7 - 7-28 When the following reaction reached...Ch. 7 - 7-29 The following reaction was allowed to reach...Ch. 7 - Prob. 7.30PCh. 7 - 7-31 Here are equilibrium constants for several...Ch. 7 - 7-32 A particular reaction has an equilibrium...Ch. 7 - Prob. 7.33PCh. 7 - Prob. 7.34PCh. 7 - 7-35 A reaction has a high rate constant but a...Ch. 7 - 7-36 Complete the following table showing the...Ch. 7 - Prob. 7.37PCh. 7 - Prob. 7.38PCh. 7 - Prob. 7.39PCh. 7 - 7-40 Is there any change in conditions that change...Ch. 7 - 7-41 The equilibrium constant at 1127°C for the...Ch. 7 - Prob. 7.42PCh. 7 - 7-43 (Chemical Connections 7A and 7B) Why is a...Ch. 7 - Prob. 7.44PCh. 7 - 7-45 (Chemical Connections 7C) A painkiller—for...Ch. 7 - 7-46 (Chemical Connections 7D) What reaction takes...Ch. 7 - Prob. 7.47PCh. 7 - Prob. 7.48PCh. 7 - Prob. 7.49PCh. 7 - 7-50 Draw an energy diagram for an exothermic...Ch. 7 - Prob. 7.51PCh. 7 - Prob. 7.52PCh. 7 - Prob. 7.53PCh. 7 - Prob. 7.54PCh. 7 - Prob. 7.55PCh. 7 - Prob. 7.56PCh. 7 - 7-57 Write the reaction to which the following...Ch. 7 - Prob. 7.58PCh. 7 - Prob. 7.59PCh. 7 - Prob. 7.60PCh. 7 - Prob. 7.61PCh. 7 - Prob. 7.62PCh. 7 - Prob. 7.63PCh. 7 - 7-64 As we shall see in Chapter 20, there are two...Ch. 7 - Prob. 7.65PCh. 7 - Prob. 7.66PCh. 7 - Prob. 7.67PCh. 7 - Prob. 7.68PCh. 7 - 7-69 Pure carbon exists is several forms, two of...Ch. 7 - Prob. 7.70PCh. 7 - 7-71 You have a beaker that contains solid silver...Ch. 7 - Prob. 7.72PCh. 7 - Prob. 7.73PCh. 7 - Prob. 7.74PCh. 7 - Prob. 7.75PCh. 7 - Prob. 7.76PCh. 7 - Prob. 7.77PCh. 7 - Prob. 7.78PCh. 7 - Prob. 7.79PCh. 7 - Prob. 7.80PCh. 7 - Prob. 7.81PCh. 7 - 7-82 An equilibrium mixture of O2, SO2, and SO3...Ch. 7 - Prob. 7.83PCh. 7 - Prob. 7.84P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7-10 The rate of disappearance of HCI was measured for the following reaction: The initial concentration of HCI is 1.85 M. Its concentration decreases to 1.58 M in 54.0 min. What is the rate of reaction?arrow_forwardHome work (1) Derive the equation for the rate of [NO2] formation for the following reaction 1 NO2 Cl →A NO2 + Cl2arrow_forwardQuestion 7 A graph of Z concentration during a chemical reaction is shown. Determine the average rate Z is being lost or gained during the first 60 seconds of reaction. -3.5e-3 M/s The instantaneous rate of Z loss/gain at 60 seconds is slower than average rate loss/gain during the first 60 seconds. [z] 0.25 0.2- 0.15- 0.1- 0.05- 0- 20 40 60 80 100 120 140 time (s) Il Proctorio is sharing your screen. Stop sharing Hide itsarrow_forward
- QUESTION 8 What is the order of the reactant when a plot of the natural log [reactant] vs time is linear? O -2 O 2 O 1/2 O 1arrow_forwardQuestion 15: For the reaction 2NOCI -> 2NO + CI2, a plot of 1/[ NOCI] vs. time in seconds yields a straight line with slope 2E-3. What is the rate law ? O rate = 4E-6 /s [NOCI] O rate=2E-3/s [NOCI] rate = 4E-6 L/mol-s [NOCI]² O rate=2E-3/s [NOCI]² rate=2E-3 L/mol-s [NOCI]²arrow_forwardQuestion 10 For the reaction A+B+CD+E, the initial reaction rate was measured for various initial concentrations of reactants. The following data were collected: Trial 1 2 3 4 [A] [B] [C] Initial rate (M) (M) (M) (M/s) 0.40 0.40 0.40 1.2x10-4 0.40 0.40 1.20 0.80 0.40 0.40 0.80 0.80 0.40 3.6x10-4 4.8x10-4 4.8x10-4 k 1.9x10 M4.8 Submit ✓ Correct Part E Previous Answers Given the data calculated in Parts A, B, C, and D, determine the initial rate for a reaction that starts with 0.65 M of reagent A and 0.80 M of reagents B and C? Submit Express your answer to two significant figures and include the appropriate units. Indicate the multiplication of units explicitly either with a multiplication dot or a dash. ► View Available Hint(s) Value Provide Feedback μA Units MacBook Pro P Pearson 10 of 15 www. res Review | Constants I Periodic Table ? Copyright © 2023 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy | Permissions | Contact Us | Next >arrow_forward
- At a certain concentration of reactants the rate of appearance of water in the following reaction is 0.21 M/s. HINT: Watch the sign of the rate! 4NH3 + 7O2 4NO2 + 6H2O (A) What is the rate of reaction of O2? ______ M/sarrow_forwardthe reaction profile is shown below 2A + B + C → G. What is the rate law for this reaction Potential Energy for the reaction A+B 12 Reaction Progress a) Vo = K[c] b) Vo = K[A] [B] [C] overall Chaose g answer from the ones given below our c) Vo =K[A] [B] d] Vo = K[A]5arrow_forwardStep 1) X(g) + Y(g) A(g) fast, reaches equilibrium (For Step 1, k1 = forward rate constant & k-1 = reverse rate constant.)Step 2) A(g) + Z(g) B(g) slow (k2 = rate constant) Step 3) B(g) P(g) fast (k3 = rate constant) a) What is the overall reaction? b) What is the rate law supported by this mechanism? Explain your answer. c) The following is experimental data found for this reaction: Initial [X] (M) Initial [Y] (M) Initial [Z] (M) Initial Rate (M/day) Exp 1 0.150 0.150 0.150 0.569 Exp 2 0.250 0.150 0.150 0.948 Exp 3 0.250 0.300 0.150 3.793 Exp 4 0.300 0.350 0.200 6.196 i) What is the experimentally determined rate law? ii) Determine the rate constant and include units. d) Does your experimentally determined rate law support the proposed mechanism? Explain your answer. e) ΔΔHo = -347 kJ/mol for the overall reaction. Draw a reasonable reaction profile for the proposed mechanism. Label your profile with the proper reactants,…arrow_forward
- Current Attempt in Progress The reaction 2A + 2B M+Nhas the rate law: Rate = k[A]?. At 25°C, k = 0.0376 L mol1s1. If the initial concentrations of A and B are 0.387 M and O.459 M, respectively Your answer is correct. (a) What is the half-life of the reaction? 68.7 seconds Hint Your answer is incorrect. (b) What will be the concentrations of A and B after 30.0 minutes? [A] = i ! M [B] = i M Hintarrow_forwardTest time left: 01:49: Н, + 1 2HI The reaction kinetics data for the above reaction is as follows. Experiment [H2] Initial rate 0.32 0.25 2.1x10-3 0.32 0.5 4.2x10-3 3 0.64 0.25 8.4x103 Write the rate law for the above reaction and find the order of the reaction with respect to individual reactants as well as order for the overall reaction. (5)arrow_forwardREACTION A +B + C Products [B](M) 1.0 2.0 1.0 1.0 Rate(M/s) 1.0E-4 4.0E-4 2.0E-4 2.0E-4 Trial 1 [A](M) [C](M) 1.0 0.010 1.0 0.010 3 1.0 0.020 2.0 0.010 Using the data in the table and the rate law below, identify the reaction order with respect to each reatant. Rate = k[A]M[B]^[c]P (Rate = k times [A] to the mth power times [B] to the nth power times [C] to the pth power) %3Darrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY